Water — Four Quiet Miracles
We recently took journeys back through the ages, from the time of primordial Earth, though the ascent of civilization, and to hypothetical futures our species may take. The star of that journey, is water.
When I think of it – there are four quiet miracles of water, in how it sustains life.
Life does not merely exist in water, but it depends on the unique properties of water itself.
This first quiet miracle is the amazing ability for water molecules to dissolve stuff and move it around, not just at a cellular level, but in the world at large. It is said that water is ‘the universal solvent’.
To understand what that means, and why, we need to zoom in closely at our star, the humble water molecule, H2O – two atoms of hydrogen bonded to an atom of oxygen.
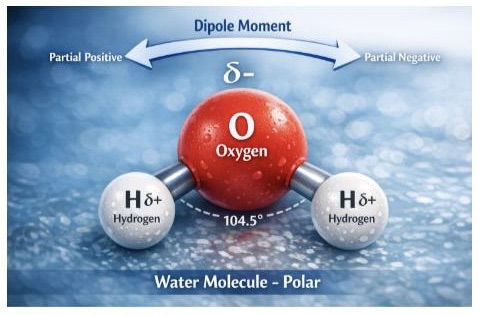
At the molecular level, a water molecule is unbalanced. Its oxygen atom pulls electrons more strongly than its two hydrogens, giving the molecule a slight negative charge on one side and a slight positive charge on the other. In chemistry, water is referred to as a polar solvent.
This polarity turns every droplet into a tiny magnet. What this does is to allow water to make salts separate, minerals to dissolve, gases to disperse, and nutrients to travel. These are all things needed for cells to work, and to support life.
Because of this asymmetry, biology quite literally, becomes possible.
A key part of this miracle is that living things need tiny charged hydrogen particles (H⁺ ions) and electrons to create energy. Inside cells, especially in mitochondria (in animals) and chloroplasts (in plants), these particles are moved across a membrane to create an energy difference, similar to water building up behind a dam. An enzyme called ATP synthase acts like a turbine, using that energy flow to make ATP. ATP is the body’s energy “currency,” much like money we use to buy things — every cell uses ATP to power movement, repair, growth, and basic survival. Just as cars run on gasoline, all living things run on ATP. Those H⁺ ions that make this happen come from water.
Inside every cell, water acts as the great meeting place, the place where cellular chemistry happens. Without water’s ability to dissolve and transport, metabolism would grind to a halt. Life’s chemistry simply does not happen without water.
The ability of water to dissolve minerals so easily, also allows for its flow to carve creeks and rivers out of sand, clay and even solid rock, allowing for the movement of life-giving water across thousands of kilometers of otherwise barren lands. Where water flows, so goes life.
The second quiet miracle is how waters density changes when it changes from a liquid, to a solid, and it reveals this each winter.
Most substances become denser as they freeze, but water does the opposite. When it solidifies, its hydrogen bonds lock molecules into a crystalline lattice that takes up more space. It’s still the same amount of water, but in a slightly greater volume. Liquid water has a density of 1.00 g/mL, while ice has a density of 0.92 g/mL – it’s lighter, and that’s why ice floats.
That quirky property of water reshapes the planet.
Bodies of water, such as lakes, rivers and oceans, freeze from the top down, forming a lid of insulation that traps liquid water beneath. Beneath winter’s glass, fish continue to swim, insects overwinter in the mud, and microbial life hums along in darkness. Were ice heavier than water, lakes would freeze solid year after year — and freshwater ecosystems would collapse. The survival of life in cold climates rests on this fact.
The third miracle lies in water’s memory of heat.
Water absorbs enormous amounts of energy before its temperature changes. It warms slowly and cools slowly, buffering both organisms and climate from violent swings. Oceans temper continents. Lakes soften winters and delay springs. Within our own bodies, blood plasma stabilizes temperature even as muscles burn and air chills.
You can think of water as a ‘heat battery’. It takes a lot of energy to warm water appreciably, but once ‘charged’, it holds that thermal energy and slowly releases it, like a battery discharging. This property of water acts as a thermal shock absorber for the planet, quietly preventing the planet from violently lurching between extremes. This ability to cushion thermal swings allows ecosystems to slowly adapt as the seasons change.
The fourth miracle is visible everywhere, though rarely noticed.
Water molecules cling to one another through cohesion, forming droplets and streams. At the same time, they adhere to other surfaces — soil particles, rock faces, the microscopic tubes inside plant stems. Together, these forces allow water to defy gravity, climbing from roots to leaves in towering trees, hundreds of feet tall. This is through a process called capillary action. These forces also allow rain to soak into fields instead of racing away.
From cloud to creek, from cell to canopy, water connects the biosphere through invisible threads of attraction.
When we look at these four quiet miracles, polarity, floating ice, thermal stability, molecular cohesion, and consider them as a whole, they form the framework upon which the entire biosphere rests. And of course, our civilization sits within that biosphere, a part of it and reliant on its predictability.
And because water just performs these quite miracles day in and day out, whether we are asleep or awake, it’s easy to take it all for granted, at least until we are reminded that the same substance capable of such generosity carries within it an equal capacity for harm – a topic for our next discussion.
References
https://watermanagement.yale.edu/topics/water-science/water-as-a-molecule/
https://www.unr.edu/nevada-today/news/2023/atp-why-is-water-unique
https://courses.ems.psu.edu/earth111/node/842
https://www.encyclopedia.com/science/news-wires-white-papers-and-books/climate-moderator-water
https://www.usgs.gov/water-science-school/science/capillary-action-and-water







